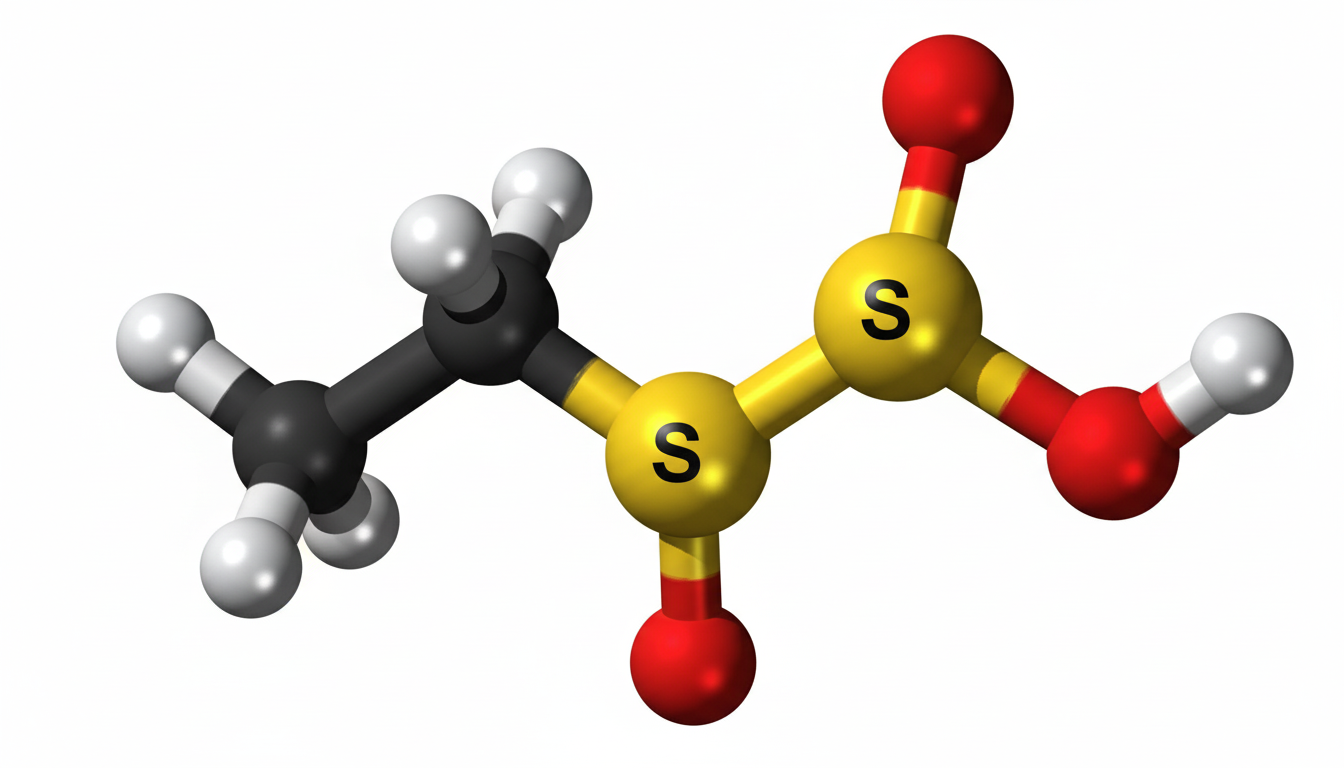
Ethylene sulfate is an important chemical compound with a wide range of applications in industrial and scientific fields. Its unique chemical structure makes it highly reactive and valuable in chemical synthesis. This compound is primarily used as an intermediate in the production of various chemicals, including surfactants, polymers, and pharmaceuticals. Understanding ethylene sulfate is crucial for chemists and industry professionals due to its reactivity and potential health impacts.
Chemical Structure and Properties
Ethylene sulfate is a cyclic ester of sulfuric acid and ethylene glycol. Its molecular formula is C2H4SO3, and it appears as a colorless to pale yellow liquid under standard conditions. It is highly soluble in water and polar organic solvents, making it versatile for chemical reactions. The compound is known for its high reactivity, particularly in nucleophilic substitution reactions. Its chemical properties, such as polarity and reactivity, allow it to serve as a building block in complex chemical processes.
Industrial Applications
Ethylene sulfate is widely used in the chemical industry for producing ethoxylated compounds. One of its primary applications is in the manufacture of surfactants, which are essential in detergents, shampoos, and cleaning agents. In the pharmaceutical industry, ethylene sulfate serves as an intermediate in synthesizing drugs and medicinal compounds. Additionally, it is employed in polymer chemistry to modify polymers for increased solubility and stability. Its versatility makes it a valuable component in multiple industrial sectors.
Role in Chemical Synthesis
The compound’s reactivity allows it to participate in a variety of chemical reactions. Ethylene sulfate reacts with nucleophiles, such as alcohols and amines, to produce ethoxylated products. These reactions are important in creating surfactants with specific properties, including foaming ability, solubility, and biodegradability. In organic synthesis, ethylene sulfate is also used to introduce ethylene oxide groups into molecules, which can significantly alter chemical behavior and performance in industrial applications.
Health and Safety Considerations
Ethylene sulfate is a hazardous chemical and must be handled with care. Direct exposure through inhalation, skin contact, or ingestion can lead to irritation, respiratory issues, and more severe health effects. In laboratories and industrial settings, strict safety protocols are followed, including the use of gloves, protective clothing, and adequate ventilation. Awareness of its toxicity and potential risks ensures safe handling and reduces the likelihood of accidents.
Environmental Impact
The production and disposal of ethylene sulfate require careful management due to its potential environmental effects. If released into water systems, it can cause contamination and harm aquatic life. Industrial facilities often implement wastewater treatment and containment measures to prevent environmental damage. Additionally, chemists are exploring greener synthesis methods to reduce hazardous byproducts, contributing to more sustainable chemical practices.
Storage and Handling Guidelines
Proper storage of ethylene sulfate is essential for safety. It should be kept in tightly sealed containers, away from heat, sparks, or incompatible chemicals. Temperature control is important to prevent decomposition or accidental reactions. In case of spills, immediate neutralization and cleanup procedures are followed to minimize risk. Adequate training for personnel working with this compound ensures safe and efficient handling in any chemical facility.
Conclusion
Ethylene sulfate is a versatile and highly reactive chemical with significant industrial and scientific value. Its role in producing surfactants, polymers, and pharmaceuticals highlights its importance in modern chemistry. However, due to its hazardous nature, proper handling, storage, and safety measures are critical. Understanding both the benefits and risks of ethylene sulfate allows chemists and industry professionals to use it effectively while minimizing environmental and health impacts.